The embryo biopsy is a procedure where a small sample of cells is removed from the embryo to test for genetic defects.
This article discusses when you should get an embryo biopsy and how it works.
If you’re thinking about doing IVF, it is essential to know this information before making any decisions!

What is an embryo biopsy?
An embryo biopsy is a procedure where five to 10 cells are removed from an early-stage embryo to analyze its DNA.
The cells removed for genetic analysis come from the trophectoderm (the outer layer of cells) that will ultimately make up the placenta.
Why do an embryo biopsy?
The reason to biopsy an embryo is to screen for genetic disorders. The most common test is preimplantation genetic testing for aneuploidy (PGT-A). (Formerly called PGS, or preimplantation genetic screening).
PGT-A looks at whether or not the embryo has a normal number of chromosomes (46). Having a numerical chromosomal abnormality is called aneuploidy.
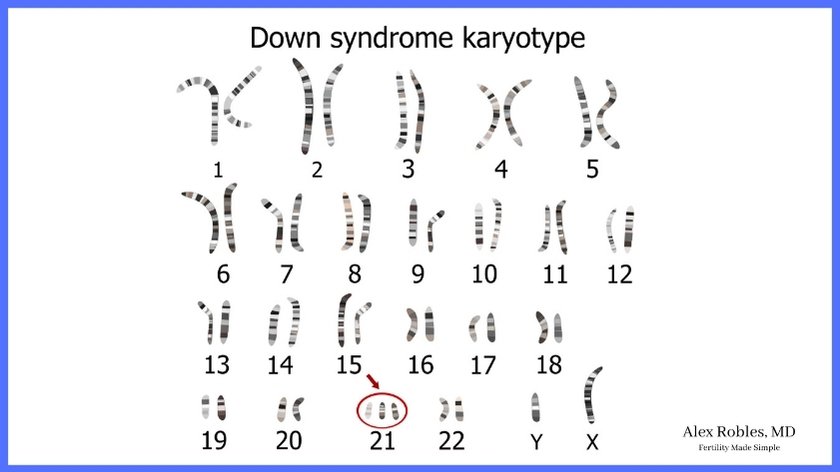
Embryo biopsies can also look for specific genetic conditions like:
- Cystic fibrosis,
- Tay Sachs disease,
- Sickle cell anemia,
- Thalassemia,
- BRCA,
- or any other genetic disorder that may run in your family.
This is called PGT-M, or preimplantation genetic testing for monogenic disorders. (Formerly called PGD, preimplantation genetic diagnosis).
The purpose of PGT-M is to transfer only unaffected embryos. We cannot change the genetics of an embryo if they have the condition.
Lastly, a biopsy can look for structural defects in the chromosomes, such as a chromosomal translocation or an inversion.
This is called PGT-SR, or preimplantation genetic testing for structural rearrangements.
Does the biopsy harm the embryo?
If performed on a blastocyst, the embryo biopsy procedure is generally considered safe. It doesn’t seem to have any apparent detrimental effects on the developing embryo.
However, the biopsy may damage the embryo if too large a sample is taken.
Overall, more research is needed on long-term effects.
It is important to note that the biopsy is taken from cells that will become the placenta.
When is an embryo biopsy done?
Typically, the embryo biopsy is done on days 5, 6, or 7 of development, when the embryo has reached the blastocyst stage.
We do not recommend having an embryo biopsy done on day 3, called a blastomere biopsy.
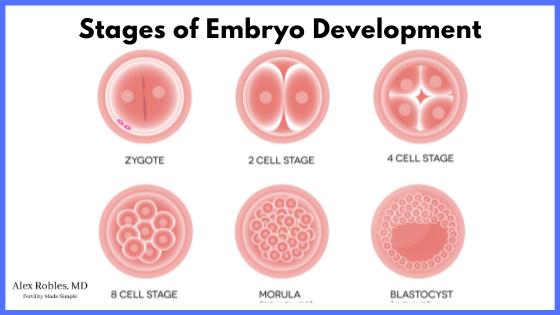
Why is an embryo biopsy done at this time?
It is when an embryo has reached the blastocyst stage that it is safest to perform the procedure.
At the blast stage, you can distinctly see the cells that will become the placenta (the outer trophectoderm cells) versus the cells that will become the baby (the inner cell mass).
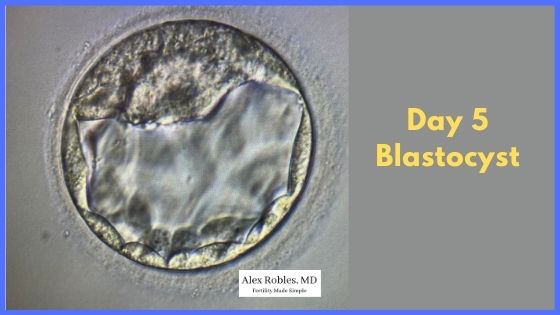
In addition, a blastocyst is composed of at least 50-100 cells.
Before the improvement of embryo culture technology, biopsies were typically done on day 3 (the cleavage stage biopsy).
How long does an embryo biopsy take?
The embryo biopsy procedure takes about 15 minutes. However, the results usually take about two weeks to return.
Who can do the embryo biopsy?
A skilled embryologist in the IVF lab is the one who performs the embryo biopsy.
How do you do an embryo biopsy?
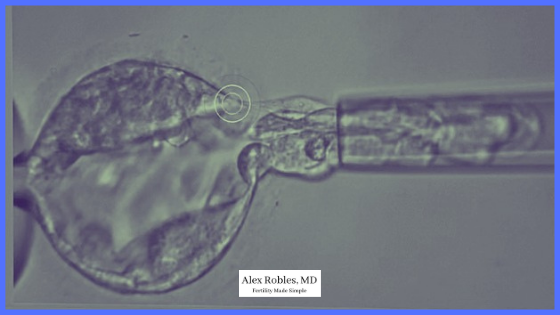
First, the embryologist uses a laser to make a small hole in the “shell” of a Day 3 embryo. This is called assisted hatching.
When the embryo makes it to the blast stage (on Day 5, 6, or 7), some cells should start hatching out of the tiny hole.
The embryologist then gently pinches off 5-10 cells that are poking out using gentle suction.
Laser pulses are then used to remove the cells from the embryo.
What happens after embryo biopsy?
The biopsy is frozen, and the embryologist sends the samples off to a lab where technicians analyze the DNA.
The embryo is also frozen as it could take anywhere between 10-14 days before the result comes back.
The laboratory looks at various characteristics of the chromosomes to look for any genetic abnormality. Not all embryos that look normal on visual inspection have a normal chromosomal makeup.
Are there any other embryo biopsy risks?
The risks of a blastocyst biopsy are usually considered very low.
A possible risk is that the embryo will not survive the procedure, though there are no large-scale studies to give a percentage.
Other risks include the possibility of a false positive or a false negative result. This means that the laboratory may say that everything looks OK, but the embryo could be abnormal (false negative).
Alternatively, they could say that there are abnormal cells when the embryo is normal (false positive).
False positives and false negatives are reported to be about 4%.
Who benefits the most from PGT-A?
One large randomized controlled trial found that PGT-A did not improve pregnancy outcomes in all women compared to un-biopsied embryos.
Biologically, this makes sense as the likelihood of making aneuploid embryos increases with age.
With that said, it is essential to note that this study only included “good prognosis patients.” In other words, these patients made multiple embryos following their in vitro fertilization cycle.
Other Related Questions
Can embryo biopsy allow for gender selection?
Yes, embryos can be biopsied to determine the gender of the embryo. We cannot change the gender; we can only tell you if it has two X chromosomes (female) or an X and Y chromosome (male).
Why are all my embryos abnormal?
Embryo aneuploidy increases with maternal age and is the leading cause of implantation failure and pregnancy loss.
Unfortunately, the likelihood of making a normal embryo decreases with age. Approximately 60-80% of embryos will be abnormal in women older than 40 years of age.
This recent study looked at the chances of making abnormal embryos based on age:
- Age 35-37: ~50% chance of making a euploid
- Age 38-40: ~38% chance
- Age 41-42: ~26% chance
Can you biopsy Day 3 embryos?
Day 3 biopsies are not recommended because evidence shows the blastomere biopsy can damage the embryo.
In contrast, no difference in implantation potential was seen in blastocyst biopsies.
How much does an embryo biopsy cost?
You can expect to pay an additional ~$3,000-5000 to do an embryo biopsy during your IVF cycle.
What is the difference between embryo biopsy vs PGT
The purpose of an embryo biopsy is to perform preimplantation genetic testing (PGT).
PGT is a technique used to screen embryos for genetic abnormalities and avoid transferring an abnormal embryo.
What is a polar body biopsy?
A polar body is formed when an egg cell goes from 46 chromosomes to 23 chromosomes. The egg does this because it needs to accept 23 chromosomes from the sperm.
A polar body biopsy removes the polar body from the egg to test it for any chromosomal abnormalities. The 23 chromosomes in the polar body should be representative of the 23 chromosomes remaining in the egg cell.
The issue is that a polar body biopsy only contains genetic information from the maternal side.
You don’t know if the sperm has the correct number of chromosomes.
Conclusion
The embryo biopsy procedure is a common add-on to IVF cycles to help select the single best embryo to transfer back into your uterus.
It is also a great way to detect the presence of a known genetic disease that runs in your family.
You should now have a better understanding of the risks and benefits of the procedure and can make an educated choice on how to approach your IVF cycle.
Be sure to speak with your doctor to learn if this is right for you.
Related Articles:
- Embryo Development: How Many Blastocyst Stage Embryos Can You Expect?
- Preimplantation Genetic Screening: What The Results Signify
- The Frozen Embryo Transfer: What Not To Do After
Make An Appointment With Dr. Robles To Discuss Your Fertility Options Today!

Alex Robles, MD
Dr. Alex Robles is a Spanish-speaking Latino-American Reproductive Endocrinologist and Infertility specialist in New York City, and a board-certified OBGYN. He has a special interest in health, lifestyle, & nutrition. Make an appointment with Dr. Robles to discuss your fertility options today!
References:
- Cimadomo D, Capalbo A, Ubaldi FM, et al. The Impact of Biopsy on Human Embryo Developmental Potential during Preimplantation Genetic Diagnosis. Biomed Res Int. 2016;2016:7193075. doi:10.1155/2016/7193075
- Zhang WY, von Versen-Höynck F, Kapphahn KI, Fleischmann RR, Zhao Q, Baker VL. Maternal and neonatal outcomes associated with trophectoderm biopsy. Fertil Steril. 2019 Aug;112(2):283-290.e2. DOI: 10.1016/j.fertnstert.2019.03.033. Epub 2019 May 15. PMID: 31103283; PMCID: PMC6527329.
- Scott RT Jr, Upham KM, Forman EJ, Zhao T, Treff NR. Cleavage-stage biopsy significantly impairs human embryonic implantation potential while blastocyst biopsy does not: a randomized and paired clinical trial. Fertil Steril. 2013 Sep;100(3):624-30. DOI: 10.1016/j.fertnstert.2013.04.039. Epub 2013 Jun 15. PMID: 23773313.
- Neal SA, Franasiak JM, Forman EJ, Werner MD, Morin SJ, Tao X, Treff NR, Scott RT Jr. High relative deoxyribonucleic acid content of trophectoderm biopsy adversely affects pregnancy outcomes. Fertil Steril. 2017 Mar;107(3):731-736.e1. DOI: 10.1016/j.fertnstert.2016.11.013. Epub 2016 Dec 8. PMID: 27939761.
- C. Hanson, T. Hardarson, K. Lundin, C. Bergh, T. Hillensjö, J. Stevic, C. Westin, U. Selleskog, L. Rogberg, M. Wikland, Re-analysis of 166 embryos not transferred after PGS with advanced reproductive maternal age as indication, Human Reproduction, Volume 24, Issue 11, November 2009, Pages 2960-2964
- Munné S, Kaplan B, Frattarelli JL, Child T, Nakhuda G, Shamma FN, Silverberg K, Kalista T, Handyside AH, Katz-Jaffe M, Wells D, Gordon T, Stock-Myer S, Willman S; STAR Study Group. Preimplantation genetic testing for aneuploidy versus morphology as selection criteria for single frozen-thawed embryo transfer in good-prognosis patients: a multicenter randomized clinical trial. Fertil Steril. 2019 Dec;112(6):1071-1079.e7. DOI: 10.1016/j.fertnstert.2019.07.1346. Epub 2019 Sep 21. PMID: 31551155.
- Rubio C, Rodrigo L, Garcia-Pascual C, Peinado V, Campos-Galindo I, Garcia-Herrero S, Simón C. Clinical application of embryo aneuploidy testing by next-generation sequencing. Biol Reprod. 2019 Dec 24;101(6):1083-1090. doi: 10.1093/biolre/ioz019. PMID: 30721942.
- Tiegs AW, Tao X, Zhan Y, Whitehead C, Kim J, Hanson B, Osman E, Kim TJ, Patounakis G, Gutmann J, Castelbaum A, Seli E, Jalas C, Scott RT Jr. A multicenter, prospective, blinded, nonselection study evaluating the predictive value of an aneuploid diagnosis using a targeted next-generation sequencing-based preimplantation genetic testing for aneuploidy assay and impact of biopsy. Fertil Steril. 2021 Mar;115(3):627-637. DOI: 10.1016/j.fertnstert.2020.07.052. Epub 2020 Aug 28. PMID: 32863013.
